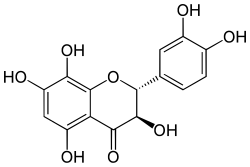
Dihydrogossypetin
 | |
| Names | |
|---|---|
| IUPAC name (2R,3R)-3,3′,4′,5,7,8-Hexahydroxyflavan-4-one | |
| Systematic IUPAC name (2R,3R)-2-(3,4-Dihydroxyphenyl)-3,5,7,8-tetrahydroxy-2,3-dihydro-4H-1-benzopyran-4-one | |
| Other names 2,3-Dihydrogossypetin 3′,4′,5,7,8-pentahydroxyflavanonol | |
| Identifiers | |
CAS Number |
|
3D model (JSmol) |
|
| ChEBI |
|
| ChemSpider |
|
PubChem CID |
|
| UNII |
|
CompTox Dashboard (EPA) |
|
InChI
| |
| |
| Properties | |
Chemical formula | C15H12O8 |
| Molar mass | 320.25 g/mol |
Except where otherwise noted, data are given for materials in their standard state (at 25 °C [77 °F], 100 kPa).  N verify (what is N verify (what is  Y Y N ?) N ?) Infobox references | |
Chemical compound
Dihydrogossypetin is a flavanonol, a type of flavonoid.[1]
Biosynthesis
The enzyme Taxifolin 8-monooxygenase hydroxylates taxifolin using NADH, NADPH, H+, and O2 to produce 2,3-dihydrogossypetin, NAD+, NADP+, and H2O.
References
- ^ Jeffrey, A. M.; Jerina, D. M.; Self, R.; Evans, W. C. (1972). "The bacterial degradation of flavonoids. Oxidative fission of the A-ring of dihydrogossypetin by a Pseudomonas sp". The Biochemical Journal. 130 (2): 383–90. doi:10.1042/bj1300383. PMC 1174417. PMID 4198081.
- v
- t
- e
Flavanonols and their glycosides
- Ampelopsin (Dihydromyricetin)
- Aromadedrin (Dihydrokaempferol)
- Dihydrogossypetin
- Dihydromorin
- Fustin (Dihydrofisetin)
- Garbanzol
- Pinobanksin
- Taxifolin (Dihydroquercetin)
- Lecontin
- (+)-fustin glucoside
- Astilbin
- Chrysandroside A
- Chrysandroside B
- Engeletin
- Eucryphin
- Smitilbin (Isoastilbin B)
- Xeractinol
 | This article about an aromatic compound is a stub. You can help Wikipedia by expanding it. |
- v
- t
- e











