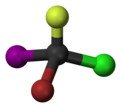
Bromochlorofluoroiodomethane
| |||
 | |||
| Names | |||
|---|---|---|---|
| Preferred IUPAC name Bromo(chloro)fluoro(iodo)methane | |||
| Other names Bromochlorofluoroiodomethane | |||
| Identifiers | |||
CAS Number |
| ||
3D model (JSmol) |
| ||
| ChemSpider |
| ||
PubChem CID |
| ||
CompTox Dashboard (EPA) |
| ||
InChI
| |||
| |||
| Properties | |||
Chemical formula | CBrClFI | ||
| Molar mass | 273.27 g·mol−1 | ||
Except where otherwise noted, data are given for materials in their standard state (at 25 °C [77 °F], 100 kPa).  N verify (what is N verify (what is  Y Y N ?) N ?) Infobox references | |||
Bromochlorofluoroiodomethane is a hypothetical haloalkane with all four stable halogen substituents present in it.[1]
Overview
This compound can be seen as a methane molecule, whose four hydrogen atoms are each replaced with a different halogen atom. As the mirror images of this molecule are not superimposable, the molecule has two enantiomers. As one of the simplest such molecules, it is often cited as the prototypical chiral compound.[2] However, since there is no synthetic route known to produce bromochlorofluoroiodomethane, the related simple chiral compound bromochlorofluoromethane is used instead when such a compound is required for research.
References

- v
- t
- e
- CH4
- CH3F
- CH3Cl
- CH3Br
- CH3I
- CH3At
- CH2F2
- CH2ClF
- CH2BrF
- CH2FI
- CH2Cl2
- CH2BrCl
- CH2ClI
- CH2Br2
- CH2BrI
- CH2I2
- CHF3
- CHClF2
- CHBrF2
- CHF2I
- CHCl2F
- C*HBrClF
- C*HClFI
- CHBr2F
- C*HBrFI
- CHFI2
- CHCl3
- CHBrCl2
- CHCl2I
- CHBr2Cl
- C*HBrClI
- CHClI2
- CHBr3
- CHBr2I
- CHBrI2
- CHI3
- CF4
- CClF3
- CBrF3
- CF3I
- CCl2F2
- CBrClF2
- CClF2I
- CBr2F2
- CBrF2I
- CF2I2
- CCl3F
- CBrCl2F
- CCl2FI
- CBr2ClF
- C*BrClFI
- CClFI2
- CBr3F
- CBr2FI
- CBrFI2
- CFI3
- CCl4
- CBrCl3
- CCl3I
- CBr2Cl2
- CBrCl2I
- CCl2I2
- CBr3Cl
- CBr2ClI
- CBrClI2
- CClI3
- CBr4
- CBr3I
- CBr2I2
- CBrI3
- CI4
 | This article about an organic halide is a stub. You can help Wikipedia by expanding it. |
- v
- t
- e
 | This article about theoretical chemistry is a stub. You can help Wikipedia by expanding it. |
- v
- t
- e
 | This stereochemistry article is a stub. You can help Wikipedia by expanding it. |
- v
- t
- e











